Your trusted partner for over 25 years.
partner for over 25 years.
Beyond Classical Boundaries
Design Safer Medical Devices with Greater Speed and Confidence
Medical device and MedTech teams must innovate quickly while meeting strict expectations for safety, reliability, usability, and regulatory readiness. From implantables and surgical tools to diagnostics, wearables, and connected devices, simulation helps engineers evaluate performance earlier—before physical prototypes reach the lab. With Ansys simulation software and SimuTech Group’s engineering expertise, teams can assess structural integrity, thermal behavior, fluid flow, RF performance, impact response, and device durability to reduce costly test cycles and make stronger design decisions.
The Medical Device Development Challenge
Advancing Safer Medical Device Innovation
Medical device teams face rising pressure to improve patient safety, prove performance, reduce testing costs, and meet strict regulatory expectations. Ansys simulation helps engineers evaluate critical design behavior earlier, supporting faster development, stronger validation confidence, and safer life-critical technologies.
Structural Integrity & Device Reliability
Ansys Mechanical
Ansys Mechanical helps MedTech engineering teams understand how device components, housings, frames, surgical tools, pumps, and implantable parts respond to real-world mechanical loads. Engineers can evaluate stress, deformation, fatigue life, vibration behavior, and material performance earlier in the design cycle—supporting safer products and more efficient verification workflows.
Key Capabilities:
- Structural FEA for device components, housings, tools, and frames
- Fatigue and durability analysis for repeated-use products
- Vibration and mechanical reliability evaluation
- Design iteration before physical prototyping
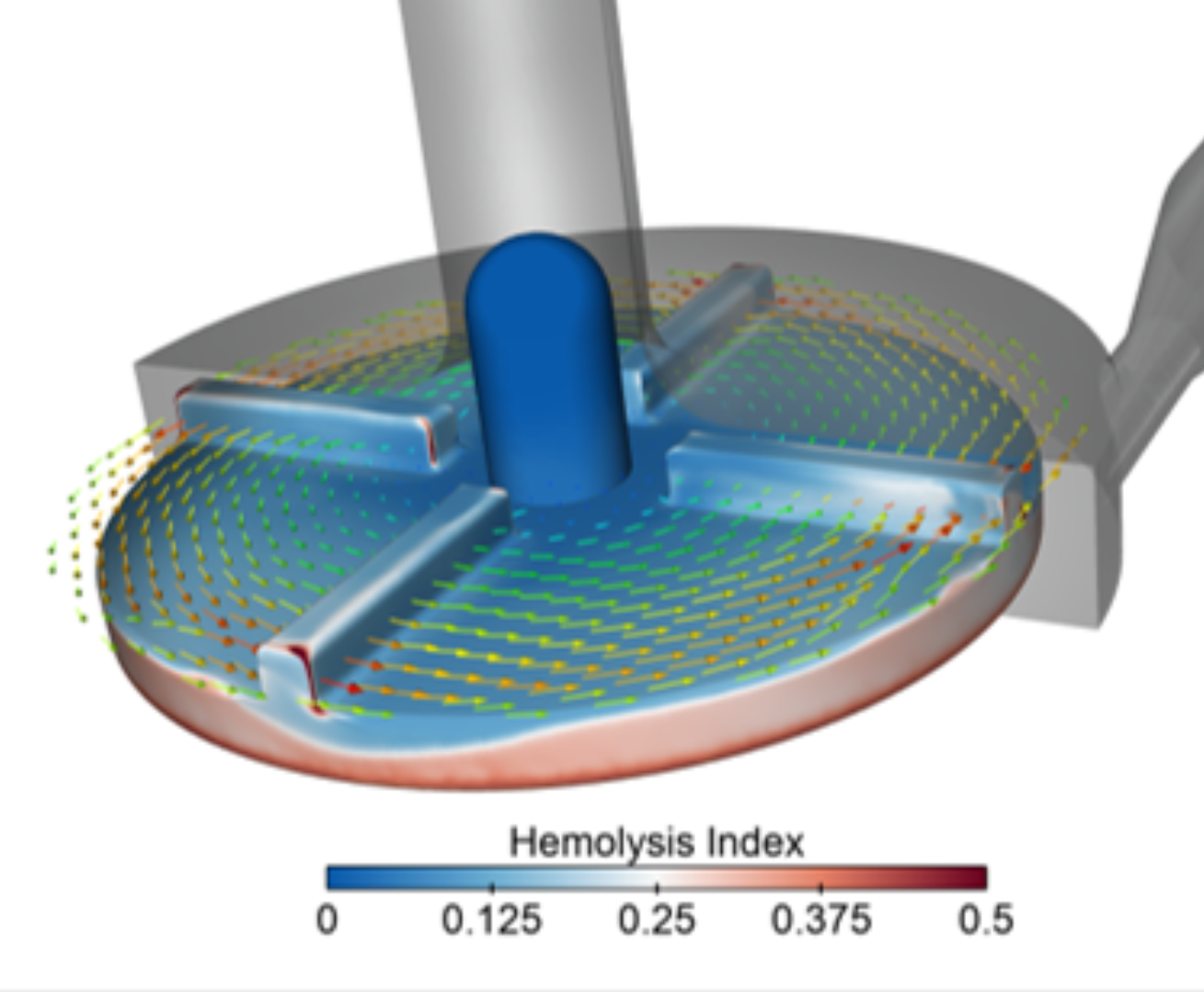
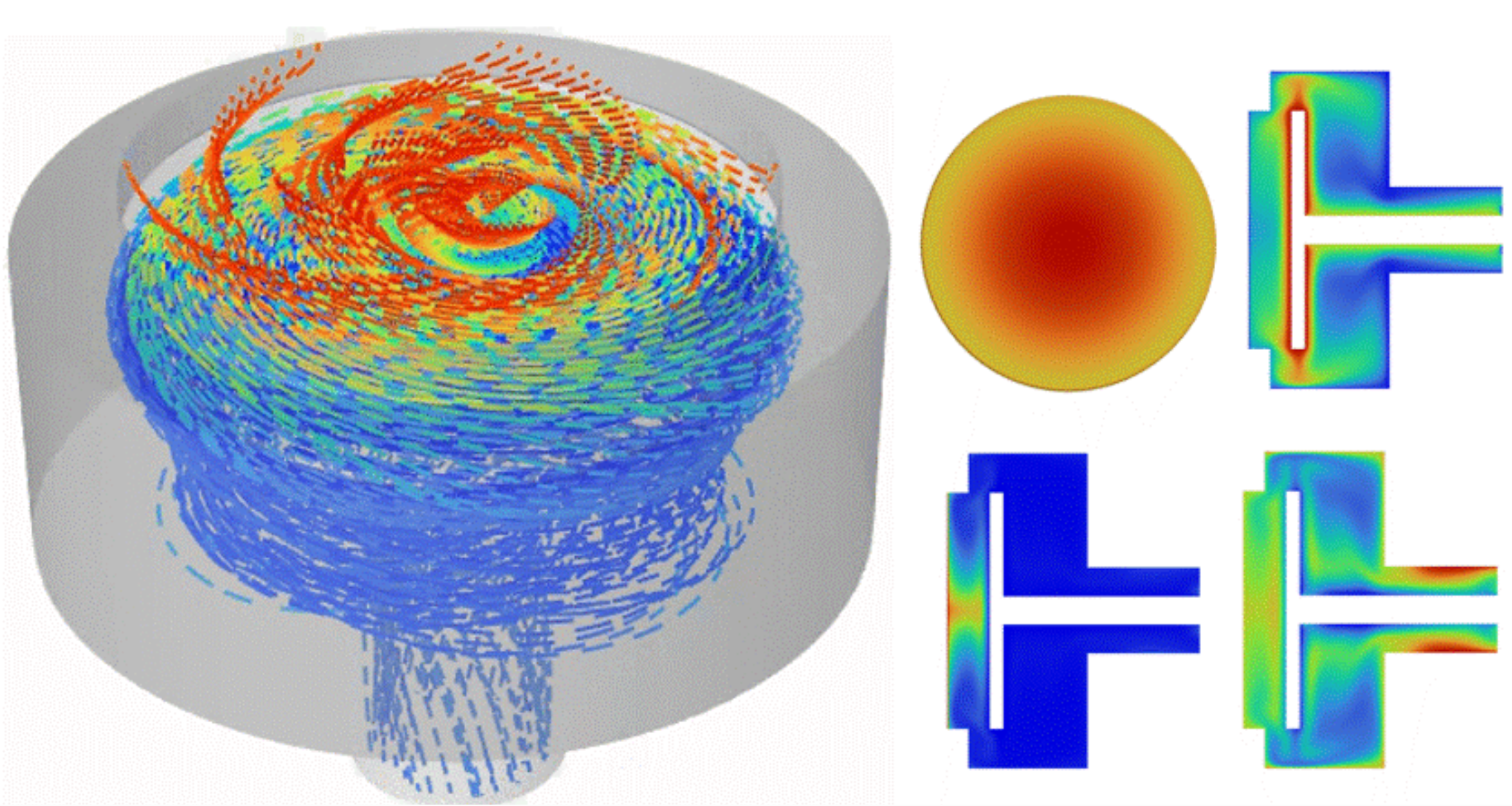
Thermal, Fluid Flow & CFD Analysis
Ansys Fluent
Ansys Fluent enables engineers to simulate fluid flow and thermal performance across a wide range of medical device applications. From blood flow and catheter behavior to electronics cooling, sterilization cycles, airflow, and temperature-sensitive device operation, Fluent helps teams identify performance risks before they become expensive testing setbacks.
Key Capabilities:
- CFD for blood flow, catheter flow, pumps, and fluid delivery systems
- Thermal analysis for electronics, sensors, and handheld devices
- Cooling strategy evaluation for diagnostic and imaging systems
- Airflow and environmental behavior modeling
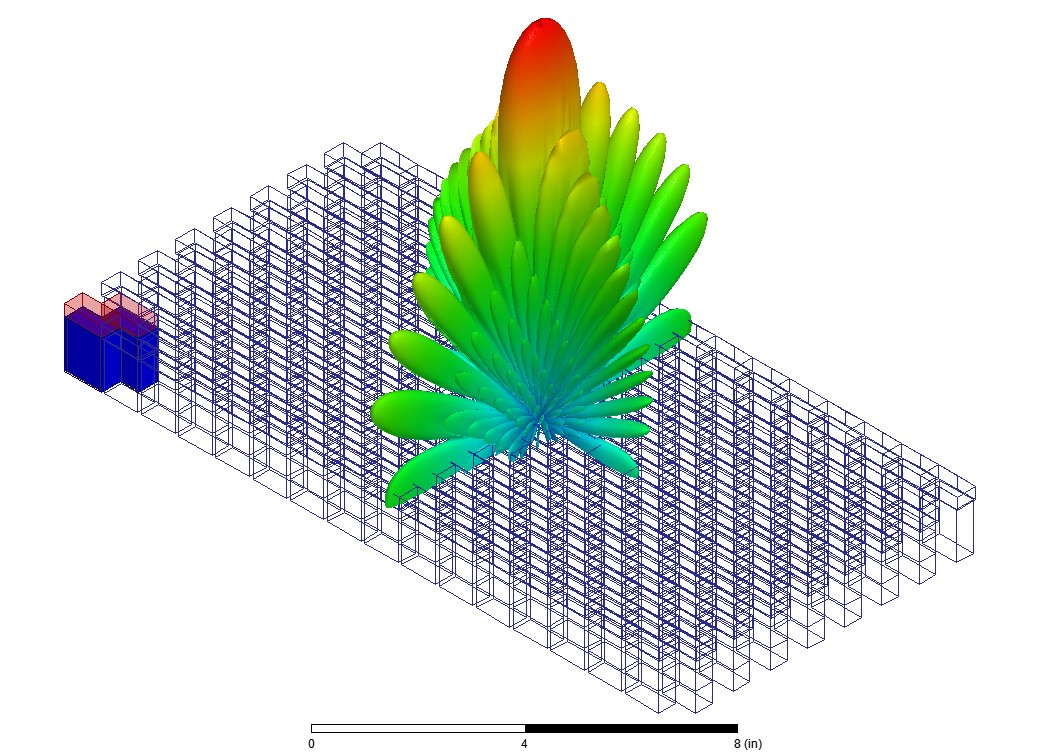
RF, Wireless & Electromagnetic Performance
Ansys HFSS
As medical devices become more connected, electromagnetic performance becomes increasingly critical. Ansys HFSS helps engineers evaluate RF behavior, antenna performance, wireless communication, EMI/EMC risk, and sensor coupling for wearables, implantables, imaging systems, and connected medical technologies.
Key Capabilities:
- RF and antenna simulation for wireless medical devices
- EMI/EMC risk evaluation
- Sensor coupling and electromagnetic interaction analysis
- Signal integrity support for connected MedTech systems
Impact, Drop & Durability Simulation
Ansys LS-DYNA
Ansys LS-DYNA gives medical device teams advanced simulation capabilities for impact, drop, fracture, shock, packaging failure, and blunt trauma scenarios. Engineers can evaluate how products respond to accidental drops, transportation loads, misuse conditions, and dynamic events—helping reduce physical testing cycles and improve product resilience.
Key Capabilities:
- Drop testing and impact simulation
- Packaging and transportation shock analysis
- Fracture, deformation, and failure prediction
- Durability evaluation for device housings and components
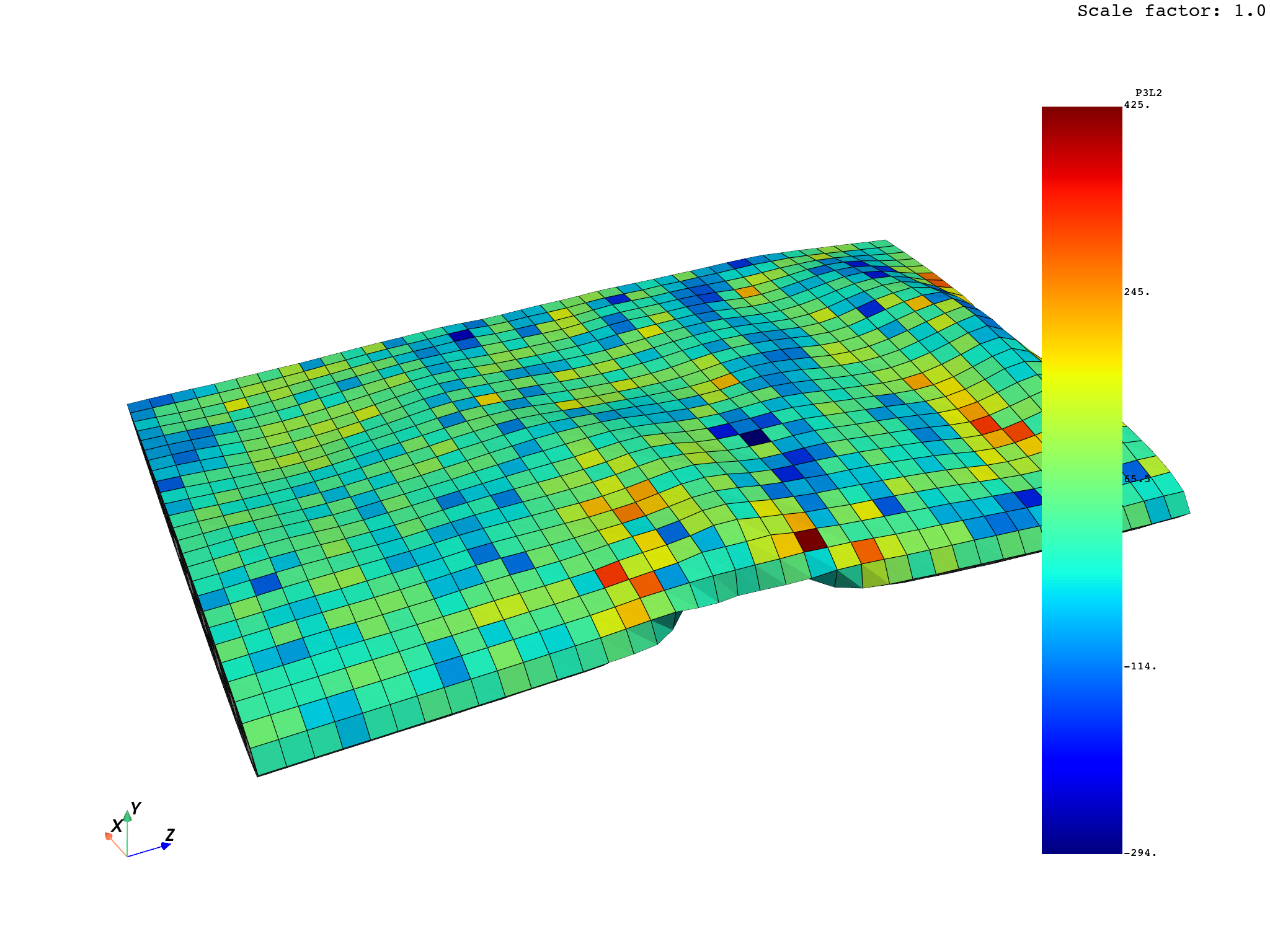
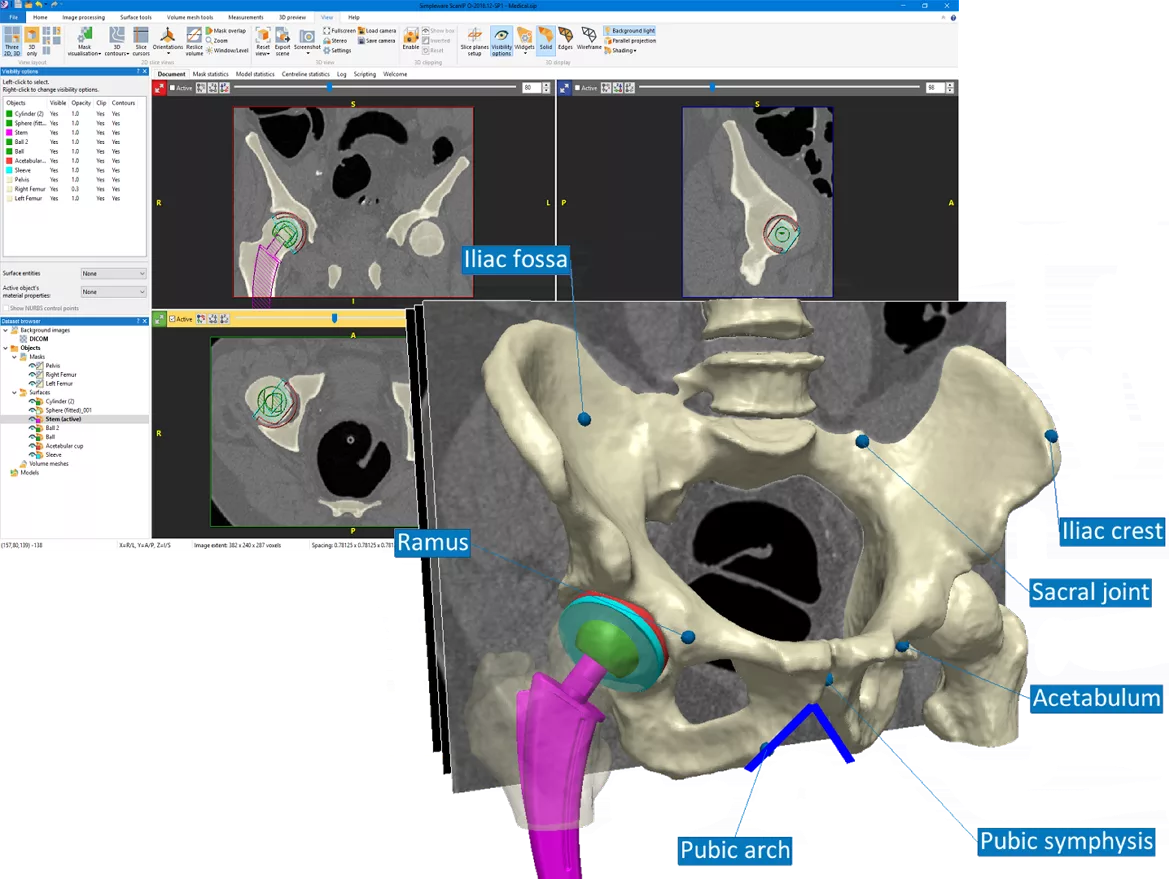
Image-Based Modeling & Simulation-Ready Geometry
Synopsys Simpleware
Simpleware software supports medical device and life sciences workflows by converting CT, MRI, micro-CT, and other 3D image data into simulation-ready models. For teams working with anatomy-specific devices, implants, surgical planning tools, or patient-derived geometries, Simpleware helps bridge the gap between medical imaging and high-quality simulation.
Key Capabilities:
- CT, MRI, and micro-CT image segmentation
- Anatomical model creation for simulation
- Mesh generation for FEA and CFD workflows
- Export to Ansys and CAD-compatible formats
Where We’ve Helped
SimuTech Group has supported engineering teams across medical device, life sciences, diagnostics, and advanced technology organizations with simulation software, consulting, training, and technical support.






Related Resources
Upcoming Webinar: Medical Device Simulation Using Ansys Fluent
Registration Coming Soon
Your Full Service Partner
Ansys Software
We are North America’s #1 Ansys partner, delivering expert sales, support, and software integration for our clients.

Simulation Consulting
We have over 25 years experience with solving complex engineering challenges with our deep simulation and domain expertise.

Training & Mentoring
We provide hands-on Ansys and simulation training led by our engineers, tailored to real-world applications and industry needs.

Physical Testing Services
We diagnose machinery faults, support failure investigations, and validate modeling parameters with advanced engineering testing.
Ready to Reduce Testing Risk and Accelerate Medical Device Development?
Whether you are improving an existing device, validating a new concept, or building a stronger simulation workflow for regulatory readiness, SimuTech Group can help. Connect with our team to explore how Ansys simulation software and expert engineering support can help you design safer, more reliable medical technologies with greater confidence.